
Dermalogica Deepens Its Aesthetics Push With FDA-Cleared Microneedling Device
Dermalogica has introduced PRO Pen Microneedling System, its first medical device cleared by the United States Food and Drug Administration, marking a significant milestone as the professional skincare brand dives deeper into advanced aesthetics.
Aurelian Lis, CEO of Unilever-owned Dermalogica, characterized the move as both an offensive and defensive investment in the professional channel as consumer-facing devices and minimally invasive treatments are proliferating rapidly. Mordor Intelligence projects the microneedling market will grow at a 10.9% compound annual rate to reach $1.1 billion by 2030, significantly outpacing broader beauty industry growth.
Lis says, “We pursued FDA clearance, along with equivalent approvals across the markets we operate in, not to look more medical, but to keep advanced treatment in qualified hands at a moment when devices are flooding the market.”
The PRO Pen is designed for licensed professionals and includes dual functionality for microneedling, a collagen-stimulating treatment that uses fine needles to create controlled microinjuries in the skin, and Luminfusion, Dermalogica’s exfoliation and infusion treatment combining nanoneedling, active ingredients and LED red light therapy. The company is emphasizing minimal downtime, expert oversight and integration with prescribed home care as key differentiators in an increasingly crowded treatment market.
While many prestige brands prioritize retail and direct-to-consumer expansion, Dermalogica is doubling down on the professional ecosystem it was built on. Aesthetician Jane Wurwand founded Dermalogica in 1986 after launching postgraduate training institution International Dermal Institute because she was dissatisfied with the skincare education available to skin therapists. Today, Dermalogica trains over 75,000 skin therapists annually, according to its website.
In the first quarter, sales in Unilever’s Beauty & Wellbeing division, which houses Dermalogica, rose 3.6%, and the company said prestige beauty growth was driven by “strong performances” from Hourglass, Tatcha and Dermalogica. In 2023, Lis told Vogue Business that a third to a half of Dermalogica’s business comes from the professional channel and about 40% of its more than 100 stockkeeping units are professional-only.
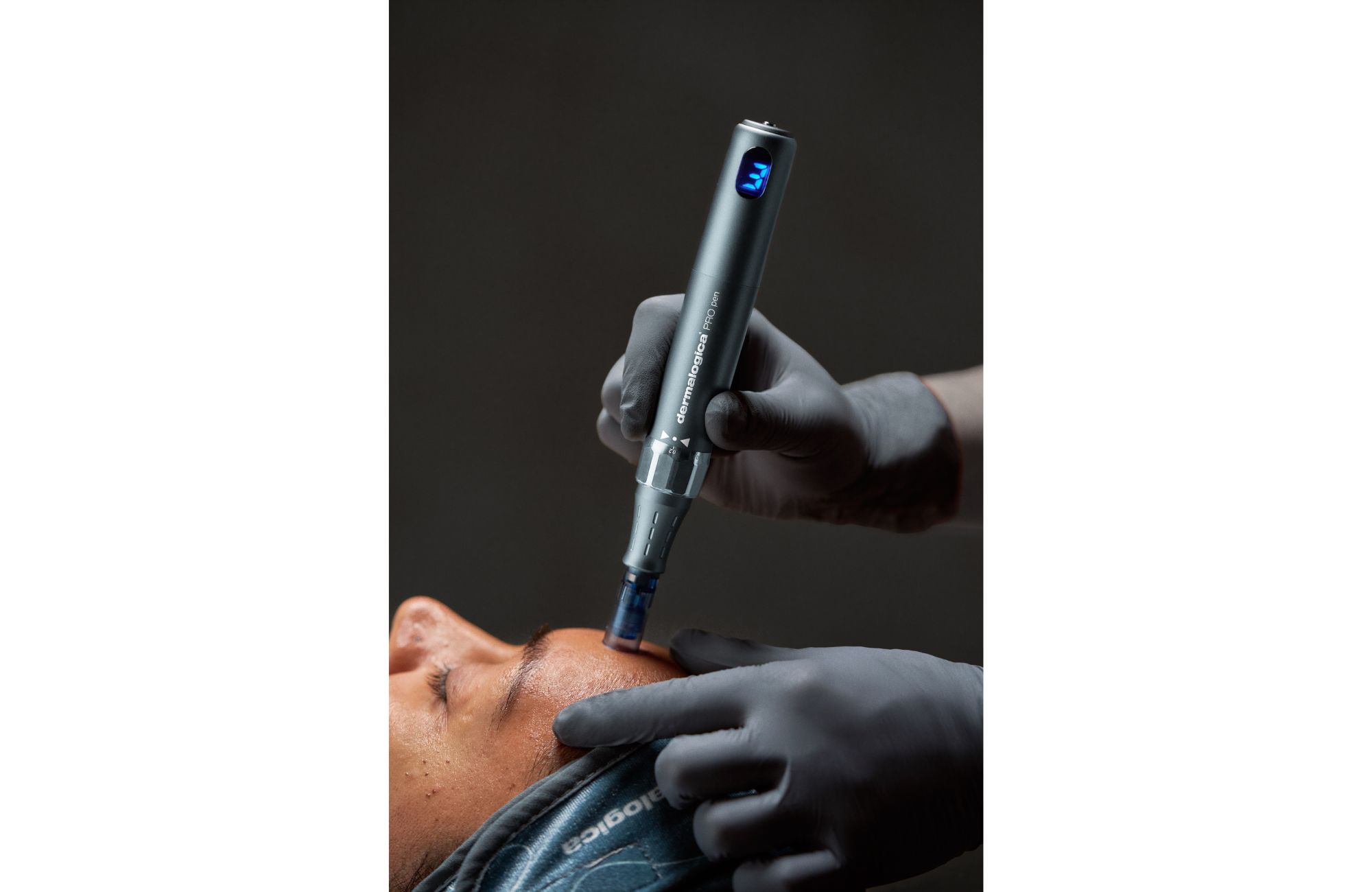
“The professional channel is the engine of our past and our future,” says Lis. “Most beauty CEOs are leaning harder into DTC and retail. We see it the other way around. Retail and DTC work because the pro channel is strong, not the reverse.”
That stance reflects broader aesthetics and skin health shifts. Treatments like microneedling, lasers and injectables are becoming mainstream, with in-office procedures being combined with ongoing product regimens. In a recent study, Boston Consulting Group and Women’s Wear Daily estimated highly engaged beauty consumers spend roughly $3,000 annually across products, procedures and longevity-focused treatments, with aesthetics commanding two-thirds of that spend to one third spent in traditional beauty categories.
Dermalogica views the integration of skincare and aesthetics as an opportunity, and it’s hardly alone in that. Obagi Medical has entered the hyaluronic acid filler field with Saypha MagIQ, and L’Oréal Groupe has secured a 20% stake in skincare and injectable giant Galderma.
Lis points to the rise of GLP-1-related skin concerns as an example of why personalized treatment plans are important. Rather than chasing quick cosmetic fixes, he argues that clients increasingly want practitioners capable of evaluating skin health holistically, including barrier health, stress, sleep and long-term maintenance.
“The treatment room is where the most advanced work happens,” he says. “The home regimen makes the gains stick. When you connect those two through a therapist who knows the client, you get better outcomes, longer relationships and a much higher lifetime value because the client keeps coming back to a person they trust.”
“The professional channel is the engine of our past and our future.”
For Dermalogica’s PRO Pen Microneedling System, FDA clearance offers reassurance to skin therapists and clients as scrutiny around aesthetic devices intensifies. The designation places the PRO Pen within a relatively limited group of FDA-cleared microneedling devices, alongside systems like SkinPen and Rejuvapen NXT, that have met the agency’s safety and performance requirements.
For the broader professional skincare space, the launch signals how legacy spas can move deeper into aesthetics without fully crossing into injectables or surgical procedures. However, Lis makes it clear that Dermalogica intends to remain focused on long-term skin health rather than structural cosmetic enhancement.
He says, “We’ll go as far into clinical as long-term skin health takes us and not a step further into volume restoration, structural distortion or any procedure that trades long-term skin integrity for a short-term look.”
The company also frames the launch as part of a broader effort to preserve the role of licensed skin therapists as technology reshapes beauty and aesthetics. As artificial intelligence-driven diagnostics and personalization tools continue to flood the market, Lis stresses that professional expertise and human relationships remain powerful assets.
“AI without a trained therapist in the room makes worse decisions, not better ones,” says Lis. “The relationship between a skin therapist and her client is the technology that matters most. AI should support that relationship, not replace it.”





Leave a Reply
You must be logged in to post a comment.